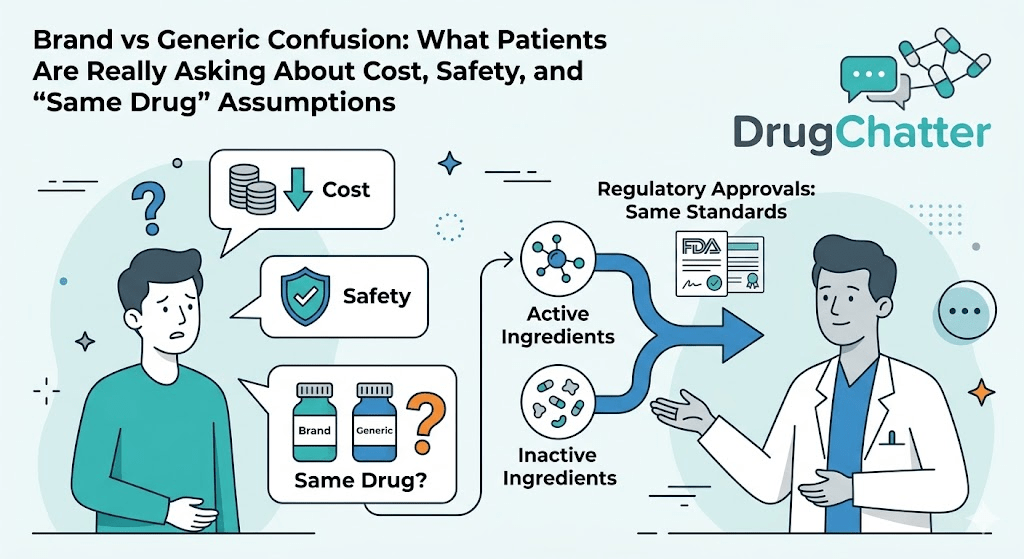
Across DrugChatter, a clear pattern emerges: patients are increasingly comfortable with switching to generics—but far less confident that “generic = identical.” The result is a steady stream of questions that blend cost concerns, safety anxiety, and misunderstandings about how branded drugs actually differ (or don’t) from their generic counterparts.
Below is a journalistic look at the most common confusion themes shaping brand vs generic drug conversations online.
1. “Is it really the same drug?” — Effectiveness skepticism around generics
The most persistent concern is whether generics deliver the same clinical effect as brand-name drugs—especially for long-established therapies like Lipitor (atorvastatin).
Questions about Lipitor dominate this category, reflecting both its legacy status and widespread generic substitution:
- Patients ask about clinical performance and perceived superiority of the brand:
https://www.drugchatter.com/chat/50179/what-studies-support-lipitor-s-superiority - Others try to reconcile generics with real-world timelines for benefit:
https://www.drugchatter.com/chat/14306/how-long-before-seeing-results-with-lipitor-alternatives - Even lifestyle factors get pulled into perceived efficacy debates, such as diet or supplements:
https://www.drugchatter.com/chat/53783/does-taking-fish-oil-reduce-the-need-for-lipitor
The underlying confusion is less about regulatory approval and more about expectation: patients often assume “brand strength” persists after patent expiry.
2. The “cheap vs safe” paradox — Online generics and trust gaps
Cost savings from generics are widely understood, but trust in sourcing is not. That gap shows up most clearly in questions about online purchasing and medication legitimacy.
- Concerns about purchasing generic Lipitor online safely:
https://www.drugchatter.com/chat/22868/is-it-safe-to-buy-lipitor-generics-online
This reflects a broader pattern: once a drug becomes generic, perceived risk shifts from the molecule itself to the supply chain. Patients trust FDA approval in principle, but worry about counterfeit risk when intermediaries enter the picture.
Manufacturing-related questions reinforce this concern even outside Lipitor:
- Regulatory oversight in controlled substances manufacturing:
https://www.drugchatter.com/chat/48297/what-safety-regulations-guide-aurobindo-s-clonazepam-manufacturing
3. Insurance, coupons, and savings confusion — “Why is brand still cheaper sometimes?”
Even when generics exist, patients frequently encounter branded drugs through coupons, rebates, or insurance incentives that blur pricing logic.
- Vascepa savings and coupon coverage questions highlight fragmented payer systems:
https://www.drugchatter.com/chat/56425/does-vascepa-coupon-cover-all-insurance-plans - Savings program mechanics add further complexity:
https://www.drugchatter.com/chat/30185/what-tools-does-vascepa-provide-for-savings-tracking - Lipitor discount programs remain a recurring point of confusion even years after generic entry:
https://www.drugchatter.com/chat/49464/are-lipitor-discount-cards-available-at-pharmacies
The paradox: a drug can be chemically generic but financially “brand-shaped” depending on how benefits are structured.
4. Side effects attribution — brand, generic, or something else?
Another major confusion driver is attribution bias: patients often struggle to determine whether side effects are caused by the active ingredient, formulation differences, or unrelated factors.
Lipitor-related discussions illustrate this clearly:
- Muscle, liver, and energy-related concerns:
https://www.drugchatter.com/chat/18159/does-lipitor-frequently-cause-breathing-difficulties
https://www.drugchatter.com/chat/21835/does-lipitor-reduce-exercise-endurance
https://www.drugchatter.com/chat/21710/how-often-do-lipitor-users-experience-increased-daytime-sleepiness
Even when generics are implied, users often still refer to “Lipitor side effects,” reflecting how brand names persist in patient cognition long after substitution.
5. Switching anxiety — “Will I feel different on the generic?”
For some medications, particularly CNS-active drugs, switching between brand and generic becomes emotionally and clinically loaded.
- Generic Xanax transition concerns:
https://www.drugchatter.com/chat/36652/what-risks-come-with-switching-to-generic-xanax - Speed of effect comparisons between brand and generic anxiolytics:
https://www.drugchatter.com/chat/33720/how-quickly-does-generic-xanax-alleviate-anxiety-compared-to-original
Similarly, questions about anticonvulsants and antidepressants reflect caution around perceived variability in response:
- Clonazepam overdose and safety concerns:
https://www.drugchatter.com/chat/20634/what-signs-indicate-a-potential-clonazepam-overdose
Even when bioequivalence is established, patient experience often drives perceived differences.
6. “Are generics just as good in complex therapies?”
In more specialized drugs, confusion shifts toward whether generics perform equally in combination regimens or hospital settings.
- Tigecycline generics vs brand equivalence:
https://www.drugchatter.com/chat/59212/are-tigecycline-generics-as-effective-as-the-brand-name - Combination therapy concerns (safety and interaction complexity):
https://www.drugchatter.com/chat/22693/are-there-any-known-risks-associated-with-lurbinectedin-combination-therapies
Here, the issue is not price—but complexity. Patients and clinicians alike question whether substitution holds under multi-drug regimens.
7. The broader market reality — patents end, perceptions don’t
Even after exclusivity ends, brand identity can continue shaping perceived value.
- Post-patent revenue dynamics for Lipitor:
https://www.drugchatter.com/chat/5094/how-does-lipitor-s-post-patent-revenue-compare-to-competitors - Competitive pricing pressures in the generic era:
https://www.drugchatter.com/chat/33629/how-does-competition-affect-lipitor-alternative-pricing - Insurance-driven savings variability:
https://www.drugchatter.com/chat/2399/how-do-insurers-formulary-designs-affect-lipitor-generic-costs
The result is a fragmented reality: chemically identical drugs competing in a market where perception, payer design, and branding still matter.
Bottom line
Brand vs generic confusion is no longer just about chemistry or regulation. It sits at the intersection of:
- patient experience
- insurance design
- digital pharmacy channels
- and the enduring psychological weight of brand names like Lipitor
The science behind generics is largely settled. The public understanding of them, clearly, is not.