How pharmaceutical companies are using AI surveillance tools to protect brand integrity, stay ahead of the FDA, and decode what patients actually say about their drugs
The call came on a Tuesday morning. A mid-sized oncology company’s regulatory affairs team was flagging a problem: an AI chatbot—one of the large consumer-facing tools used by tens of millions of Americans—was telling patients that their blockbuster therapy caused liver damage in a specific subset of patients. The claim wasn’t in the label. It wasn’t in any peer-reviewed literature the company could identify. But patients were reading it, copying it into support group forums, and citing it in conversations with their oncologists.
The company had no monitoring system in place. By the time their team discovered the misinformation, it had been circulating for six weeks.
This is no longer an edge case. As AI-generated content becomes a primary medical information layer for patients, caregivers, and even clinicians, pharmaceutical companies face a category of reputational and regulatory risk that didn’t exist three years ago. Their existing pharmacovigilance frameworks, social listening tools, and media monitoring contracts weren’t built for it. And the FDA hasn’t finished writing the rules yet—which is precisely the moment when the companies paying closest attention will determine what ‘compliance’ ends up meaning.
The Shift That Rewired the Information Stack
Before large language models became mainstream, the pharmaceutical information environment was messy but mappable. You had labeled prescribing information, DTC advertising subject to FDA review, journal articles, conference abstracts, and a long tail of social media posts that compliance teams had been monitoring, imperfectly, since roughly 2009.
Brand teams knew where the risks lived. Detailing was controllable. A medical affairs officer could read every piece of promotional material before it left the building.
What changed between 2022 and 2025 wasn’t just the introduction of ChatGPT. It was the mass adoption of AI tools as a first-stop health resource. According to a 2024 survey conducted by the Kaiser Family Foundation, nearly one in four American adults had used an AI chatbot to look up health or medical information. A separate study published in JAMA Internal Medicine found that AI chatbot responses to common patient health questions scored higher on perceived empathy than physician responses—which tells you something important about adoption dynamics even if it tells you nothing about accuracy. <blockquote> “Roughly 40% of patients now report consulting an AI tool before or after a physician visit about a new prescription, according to a 2024 Wolters Kluwer Health survey of 1,000 U.S. adults.” </blockquote>
That number is uncomfortable for regulatory teams because it means AI-generated drug information is influencing the therapeutic relationship at exactly the moment of highest leverage: when a patient decides whether to fill a prescription and whether to keep taking it.
The companies that treat this as a communications problem are behind. The ones treating it as a data and surveillance problem are starting to catch up.
What AI Actually Says About Your Drug—and Why It Matters
Pharmaceutical brand teams have spent decades understanding what physicians think about their products, what payers believe about their cost profiles, and what patients say to each other on Reddit. They have quantitative tracking studies, NPS scores, and formulary access dashboards.
None of those tools answers the question: what does GPT-4o say when a 54-year-old woman with rheumatoid arthritis asks whether she should be worried about taking your JAK inhibitor?
The answer to that question is now consequential in multiple directions.
First, there’s the regulatory dimension. The FDA has been clear, in draft guidance and in warning letters, that it considers certain types of AI-generated content to be promotional material if a pharmaceutical company had any role in generating or amplifying it. The agency’s November 2023 draft guidance on AI-generated content in promotional labeling did not resolve every ambiguity—it opened several new ones. What constitutes ‘material connection’ between an AI output and a drug manufacturer? If a company trains a fine-tuned model on its own medical affairs content, does the output require regulatory review? These questions are live. Attorneys at firms representing Pfizer, Merck, and AstraZeneca are billing hours on them weekly.
Second, there’s the pharmacovigilance dimension. The FDA’s expectations around adverse event reporting have been expanding to cover digital and social channels. In March 2024, the agency issued an update to its social media adverse event guidance that explicitly mentioned AI-generated content as a channel requiring monitoring. If your drug is being associated with an adverse event in AI outputs—even inaccurately—and you become aware of that association, you may have a reporting obligation. ‘Becoming aware’ is the operative phrase. Ignorance is a shrinking defense.
Third, there’s the brand dimension. AI tools aggregate and compress information in ways that can entrench narratives. If ChatGPT or Perplexity or Gemini consistently describes your drug as ‘less effective than competitor X’ or as carrying a risk profile that’s more severe than what the label says, that characterization will influence patient behavior, physician conversations, and ultimately market share—without a single paid advertising impression being placed.
The FDA Regulatory Exposure: What Companies Are Actually Getting Wrong
The FDA’s enforcement posture on AI-generated promotional content is still forming, but the directional signals are clear enough to act on.
The agency’s Office of Prescription Drug Promotion (OPDP) issued its first formal observation related to AI-generated promotional content in a warning letter in late 2023 to a specialty pharmaceutical company that had used a chatbot on its branded patient website. The chatbot, trained on approved promotional materials, was providing responses that OPDP determined went beyond the approved indication. The company’s position—that the chatbot outputs were dynamically generated and therefore not ‘promotional labeling’ in the traditional sense—did not hold up.
What this means operationally is that pharma companies need to know what AI systems are saying about their products on two tracks: what their own AI tools are saying (which is a compliance and MLOps problem), and what third-party AI tools are saying (which is a monitoring and intelligence problem).
The second track is where most companies have the bigger gap.
Consider what happens when a major LLM is asked about a drug’s off-label uses. Unlike a physician who makes an off-label prescribing decision within a documented clinical relationship, an AI tool might describe off-label applications to any user, without clinical context, and do so consistently at scale. If a drug company is aware that an AI system is providing this kind of information and benefits from it commercially—say, because it’s driving prescriptions—OPDP has signaled it will look at whether the company had any role in shaping that information environment.
This is not hypothetical. The FDA sent an advisory communication to three companies in Q1 2024 asking them to document their awareness of AI-generated content about their products. The companies involved have not been publicly identified, but the communication circulated among regulatory affairs professionals at major pharma companies.
Brand Share in the Age of Generative AI
Set aside regulatory exposure for a moment. The commercial stakes are straightforward.
When a patient with newly diagnosed Type 2 diabetes goes to an AI tool and asks which GLP-1 agonist is right for them, the answer they receive functions like a recommendation from a highly accessible, always-available clinician. Wegovy and Ozempic have dominated this AI recommendation landscape in a way that their share of promotional voice doesn’t fully explain—partly because of the extraordinary volume of earned media coverage around semaglutide, which AI tools trained on that corpus have absorbed and reproduced.
For competing products, this creates a measurable commercial problem. Tirzepatide (Mounjaro, Zepbound) has superior Phase 3 efficacy data on multiple endpoints versus semaglutide in head-to-head trials. But in 2024, AI tools were consistently recommending Ozempic over Mounjaro at a higher rate than the clinical evidence would predict. Eli Lilly’s medical affairs team became aware of this gap and began documenting it systematically.
The companies that built AI monitoring infrastructure early—and there are only a handful—are now able to generate something analogous to a share of voice report, but for AI outputs. They can track:
- How often their drug is mentioned in AI responses to relevant queries
- Whether the AI characterizes their drug’s efficacy as superior, equivalent, or inferior to competitors
- Whether safety signals are described accurately versus in an exaggerated or distorted way
- What patient profiles the AI recommends the drug for, and whether those profiles match the approved indication
This is brand intelligence that didn’t exist before 2023. The companies building systematic processes around it now will have a material informational advantage over those that don’t.
Voice of the Customer Has Moved—and Most Pharma Teams Missed It
There’s a parallel conversation happening at the patient level that brand teams have been slower to recognize.
For years, ‘voice of the customer’ in pharma meant patient advisory boards, focus groups, social listening on Facebook and Reddit, and occasionally mining Amazon reviews of OTC products. Those inputs are still valuable. But they capture patient sentiment after the fact—what patients say once they’ve already formed opinions.
AI query data is different. When a patient types ‘does methotrexate cause hair loss and is it permanent’ into an AI tool, that query represents an active moment of uncertainty—one where the answer they receive will shape their behavior. That query is also a signal about what’s driving anxiety, what the gaps in patient education are, and what your competitor’s patients are asking about their drugs versus what your patients are asking about yours.
The problem is that most pharma companies can’t see this data. The major AI providers don’t publish it. But there are proxy approaches: monitoring the questions patients ask in AI-enabled interfaces that companies control, auditing AI tool outputs systematically using representative patient query frameworks, and partnering with platforms that have visibility into consumer AI usage patterns.
DrugChatter, a pharmaceutical AI monitoring platform, is one of several companies that has built infrastructure specifically for this monitoring gap. The platform generates systematic audits of what major consumer AI tools say about specific drugs across hundreds of query variations—covering indication accuracy, safety characterization, competitor comparisons, and patient-population targeting. The output is a brand intelligence report that regulatory and commercial teams can use in parallel: regulatory teams get documentation of what third-party AI is saying about their drugs (important for FDA awareness obligations), and brand teams get a competitive picture of their AI share of voice.
The category is new enough that most companies are still figuring out where it lives organizationally. Some regulatory affairs teams own it. Some medical affairs teams own it. In the most sophisticated organizations, it sits in a cross-functional working group that includes regulatory, commercial, and digital health—which is probably the right model, given that the outputs matter to all three.
Case Study: How a Misrepresented Safety Signal Circulated in AI Outputs
In 2023, a branded SGLT2 inhibitor was the subject of a citizen petition to the FDA requesting a new boxed warning related to a specific cardiac event. The petition was submitted by a plaintiffs’ law firm representing patients in ongoing litigation. The petition itself was publicly available and was covered in several trade publications.
Within weeks of the petition’s filing, multiple AI tools began characterizing the drug as carrying a boxed warning for this cardiac event—even though no such warning existed or had been approved by the FDA. The AI tools were apparently synthesizing the petition coverage and legal filings with the existing label and producing a hybrid characterization that was factually incorrect.
The drug’s manufacturer had no systematic AI monitoring in place. They became aware of the problem through an unsolicited email from a cardiologist who had noticed the discrepancy while using an AI tool to look up the drug’s prescribing information for a patient.
By the time the company began auditing AI outputs and documenting the scope of the mischaracterization, it had been circulating for approximately four months. The company’s regulatory team prepared a formal record of the issue and began the process of submitting correction requests to the AI providers—a process that is labor-intensive, inconsistently successful, and doesn’t move quickly.
The commercial impact was difficult to isolate, but the company’s market research team documented a statistically significant increase in ‘unsolicited safety concern’ mentions in HCP conversations during the same quarter—a metric that typically moves when external information is shaping physician conversations in ways that aren’t driven by the company’s own communications.
This is the compounding risk of unmonitored AI environments: a factually incorrect characterization of your drug’s safety profile can propagate through the most-consulted information layer in the country, influence physician conversations, and generate adverse event reports—all before your team knows it’s happening.
Regulatory Litigation Risk: The Documentation Problem
There’s a litigation dimension that pharma legal teams are beginning to price in.
In personal injury litigation involving pharmaceutical products, plaintiffs’ counsel routinely argue that manufacturers had constructive knowledge of safety signals—meaning that even if the company didn’t know specifically, they should have known given reasonable monitoring. Courts have held companies to this standard for social media, for adverse event reports in foreign markets, and for published case reports in medical literature.
The question of whether AI-generated content creates a similar constructive knowledge standard hasn’t been litigated definitively. But several ongoing mass tort cases involving pharmaceutical products have included discovery requests specifically asking whether the company monitored AI-generated content about its products and what protocols it had in place.
A company that has no AI monitoring program produces a very different discovery record than a company that has a documented, systematic process. The absence of monitoring—particularly after industry guidance from both the FDA and industry groups like PhRMA has flagged this as an active regulatory concern—will eventually be used to argue that a company was deliberately avoiding knowledge it had an obligation to acquire.
Law firms advising pharmaceutical clients on enterprise risk are increasingly recommending that companies implement AI monitoring programs specifically because the documentation those programs generate is protective. If you can demonstrate that you were systematically monitoring AI outputs, that you identified a specific mischaracterization, and that you took documented steps to address it, your legal exposure in related litigation is materially different from the company that has nothing.
What the FDA Guidance Actually Says—and What It Doesn’t
The FDA’s regulatory framework for AI in pharmaceutical promotion is a patchwork of existing guidance applied to a new environment, with some new layers being added.
The agency’s core principle—that promotional materials must be accurate, balanced, and not misleading—applies regardless of the medium. But AI creates medium-specific problems that existing frameworks weren’t designed to address.
The 2023 draft guidance on artificial intelligence in drug promotion is worth reading carefully because it establishes several principles that will shape enforcement for years. The guidance distinguishes between AI tools that are ‘interactive promotional labeling’ (chatbots on branded websites, for example) and AI tools that are third-party systems where the manufacturer has no direct involvement. For the former category, OPDP’s expectations are essentially the same as for any other promotional material: the content needs to be reviewed, approved through the standard MLRR process, and compliant with the approved labeling.
For third-party AI, the guidance is less prescriptive but not silent. The FDA notes that companies should be ‘aware of’ AI-generated content about their products and suggests that companies with evidence of widespread AI-generated misinformation about their products should consider submitting correction requests to AI providers.
What the guidance doesn’t do is specify how frequently companies need to monitor, what methods they need to use, or what the enforcement threshold is for failing to detect AI-generated misinformation. These gaps are intentional—the FDA rarely prespecifies enforcement metrics—but they mean that companies are setting their own standards in a regulatory vacuum, which is always a risky position.
The OPDP has indicated informally through conference presentations—notably at the DIA Annual Meeting in 2024—that it views AI monitoring as part of the same broad ‘surveillance and monitoring’ obligation that covers social media. That framing is important. Social media monitoring has been an FDA expectation since the 2014 draft guidances on internet and social media platforms. Companies that framed it as optional and didn’t build systematic programs spent years playing catch-up. The FDA has never been patient about regulated industries treating its draft guidances as suggestions.
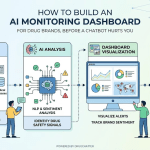
The Technical Infrastructure Behind AI Monitoring
Building an AI monitoring capability isn’t as simple as running a few queries into ChatGPT every month. The technical challenge is non-trivial for several reasons.
LLMs are probabilistic systems. A single query can produce different outputs on different days, depending on model updates, context window variations, and stochastic generation. A monitoring program that runs a handful of representative queries once a quarter will miss the variation that matters. You need systematic sampling across query types, across AI platforms, and across time—and you need a methodology for aggregating those samples into a reliable picture of what any given AI tool is likely to say about a drug under realistic patient query conditions.
The query design problem is equally complex. A patient doesn’t ask ‘what is the efficacy of Drug X versus Competitor Y on the primary endpoint of a Phase 3 clinical trial.’ They ask ‘is Drug X better than Drug Y for my condition.’ The linguistic distance between how patients actually phrase health questions and how regulatory teams think about those questions is enormous—and a monitoring program that only queries AI tools with clinical phrasing will produce a systematically biased picture of what patients are actually receiving.
DrugChatter addresses this by building query frameworks from actual patient language—sourced from forum data, patient advisory input, and social listening corpora—and running those frameworks across multiple major AI platforms simultaneously. The platform captures outputs, classifies them against a rubric that covers indication accuracy, safety characterization, competitive positioning, and patient population targeting, and generates a normalized score that brands can track over time.
The competitive intelligence application is becoming as important to brand teams as the regulatory application. If Competitor Y’s drug is being recommended by AI tools for a patient population where your drug also has approval, and you’re not monitoring that, you’re operating blind in a channel that’s now more heavily consulted than WebMD for certain demographics.
What Pharma Medical Affairs Teams Should Own
The organizational question matters as much as the technical one.
In most pharmaceutical companies, medical affairs owns the peer-reviewed literature environment, clinical data dissemination, and HCP scientific exchange. Regulatory owns promotional review, FDA communications, and label strategy. Brand owns market research, competitive intelligence, and commercial messaging.
AI monitoring cuts across all three. The content that AI tools generate about drugs is part scientific (it draws on journal articles and clinical data), part promotional (it functions like a recommendation in many contexts), and part brand intelligence (it shapes competitive positioning in the HCP and patient information environment).
The companies that have stood up AI monitoring programs report that the most effective organizational model puts medical affairs in the lead role—because medical affairs has the clinical credibility to evaluate whether AI outputs are accurate, and because they’re positioned to issue scientific corrections through appropriate channels—with regulatory providing oversight and commercial teams receiving the output as brand intelligence.
Medical affairs leadership has been slow to claim this space in some companies because it feels unfamiliar. The skill set for monitoring peer-reviewed literature and the skill set for auditing AI outputs are different. But the underlying mission—ensuring that accurate scientific information about their drugs is available to the people who make prescribing and treatment decisions—is identical.
The companies that have made AI monitoring a medical affairs priority are producing work product that’s genuinely useful across the organization: regulatory gets documentation of third-party AI behavior, commercial gets competitive intelligence, and legal gets protective records. That’s a strong ROI case for a function that has historically struggled to quantify its commercial contribution.
Adverse Event Reporting in an AI-Mediated World
The pharmacovigilance implications of AI monitoring deserve their own treatment.
Under 21 CFR Part 314 and the FDA’s adverse event reporting regulations, pharmaceutical companies are required to submit reports of adverse drug experiences when they receive information—from any source—that reasonably suggests a causal relationship between their product and an adverse event.
AI-generated content is increasingly a source of adverse event signals, in two ways.
The first is direct: patients or caregivers who describe adverse events in conversations with AI tools, and where those conversations are visible to the company through monitored channels. This is less common currently but will grow as companies deploy AI tools in patient support contexts.
The second is indirect: AI tools that consistently associate a drug with a specific adverse event—accurately or inaccurately—may be reflecting an aggregate of patient-reported information that the AI tool’s training incorporated from forums, case reports, and other sources. If a company’s AI monitoring identifies that a specific adverse event is being consistently mentioned in AI outputs about their drug, that signal warrants evaluation by their pharmacovigilance team, regardless of whether the association is accurate.
This is a genuinely novel situation for PV teams. The traditional adverse event signal sources—MedWatch, scientific literature, HCP reports, patient reports through call centers—have established workflows. ‘AI output monitoring as a PV input’ does not yet have a standardized workflow in most companies. The FDA hasn’t prescribed one. But the underlying regulatory obligation is clear enough that companies should be building one proactively.
International Regulatory Exposure: EMA, PMDA, and the Global Picture
The FDA isn’t the only agency watching this space.
The European Medicines Agency published a reflection paper on AI and machine learning in 2023 that touched on AI-generated promotional content, framing it within the existing EU pharmaceutical advertising directive (Directive 2001/83/EC). The EMA’s position—consistent with its general regulatory philosophy—is more principles-based than the FDA’s approach, but the directional expectations are similar: pharmaceutical companies should be aware of what AI systems are communicating about their products, and companies with evidence of inaccurate AI-generated content have an obligation to address it.
The PMDA in Japan has been even more explicit. In a 2024 guidance document on digital marketing in the pharmaceutical sector, the PMDA specifically listed AI-generated content monitoring as a component of compliant digital surveillance programs. Japan’s regulatory environment tends toward specificity, and the PMDA’s inclusion of AI monitoring as an explicit expectation is a signal that other agencies will follow.
For global pharmaceutical companies managing multi-market regulatory strategies, this creates a synchronization challenge. The AI monitoring infrastructure that meets FDA expectations may not meet PMDA requirements without modification, because the query frameworks, output classification rubrics, and documentation standards differ across regulatory environments. Companies building these programs now have the opportunity to design them with global regulatory harmonization in mind—which is significantly cheaper than retrofitting a US-only system later.
Competitive Intelligence: The Commercial Upside of AI Monitoring
Pharmaceutical competitive intelligence has historically operated through market research surveys, HCP interviews, conference intelligence, and secondary data analysis of claims and prescription data. These are expensive methods with significant lag time—a survey fielded in Q1 produces insights in Q2.
AI monitoring produces competitive intelligence with a latency of days, not months. And it captures a dimension of competitive positioning that traditional methods miss entirely: how the information environment is characterizing competitors.
The practical applications are significant. If an AI monitoring program reveals that a competitor’s drug is being characterized as the preferred first-line therapy in AI outputs—ahead of your drug—in a patient population where both drugs are approved, that’s an actionable commercial intelligence finding. It tells you that the AI information environment is shaped by factors that your promotional investment hasn’t yet influenced—clinical publication volume, social media coverage, KOL publishing activity, or patient advocacy communications.
Conversely, if AI monitoring reveals that a competitor’s drug is being associated with an adverse event more prominently than your label comparison would predict, that’s also useful intelligence: it may reflect emerging clinical experience, patient-reported signals in the AI training corpus, or litigation activity that’s shaping the AI’s characterization.
Brand teams that have seen this kind of AI-sourced competitive intelligence describe it as qualitatively different from what they get from traditional methods—less about stated preferences and more about the ambient information environment in which prescribing decisions are actually made.
What Good AI Monitoring Looks Like in Practice
For regulatory and commercial teams building or evaluating AI monitoring programs, the methodological requirements are becoming clearer.
Coverage needs to include at least the major consumer AI platforms—ChatGPT, Gemini, Perplexity, Claude, Meta AI—and should include the AI-assisted search features now embedded in Google and Microsoft Bing. Those search AI features reach users who would never self-identify as ‘AI tool users’ but are receiving AI-generated drug information through their normal search behavior.
Query frameworks need to be developed systematically, not ad hoc. A rigorous program will include queries across multiple patient personas, multiple query phrasings for the same underlying question, and queries designed to probe specific dimensions of concern: safety, efficacy, indication accuracy, competitive comparisons, dosing information, and patient population targeting.
Output capture needs to happen at regular intervals. Quarterly is insufficient. Model updates can change AI behavior significantly between quarters, and a single-point-in-time audit is not defensible as a surveillance program. Monthly sampling with real-time alerts for high-priority queries—like brand name plus specific safety terms—is more appropriate.
Documentation standards need to meet regulatory defensibility requirements. That means timestamped captures, reproducible methodology, and records management consistent with FDA 21 CFR Part 11 requirements if the program is being managed within a regulated system.
Escalation pathways need to be defined in advance. If an AI monitoring alert surfaces a significant inaccuracy—a safety signal characterization that doesn’t match the label, an off-label indication being recommended—the question of who evaluates it, who decides on response action, and who owns the regulatory documentation needs to be answered before the alert arrives.
The ROI Case for AI Monitoring Investment
The business case for pharmaceutical AI monitoring rests on four quantifiable risks:
Regulatory enforcement costs. A warning letter from OPDP triggers mandatory corrective advertising, pulls promotional materials from circulation, and creates reputational damage that compounds over years. Warning letters have been issued for promotional violations that were less systematic than unmonitored AI misinformation at scale. The cost of a warning letter response typically runs into seven figures when you account for legal fees, corrective campaign costs, and disruption to promotional activities.
Litigation exposure. In mass tort litigation, the difference between a company that can produce documented AI monitoring records and a company that cannot is a difference in settlement leverage that legal teams increasingly recognize. The discovery requests are coming whether or not companies are ready for them.
Market share impact. AI share of voice is becoming a quantifiable commercial metric. Companies that can demonstrate a correlation between AI output characterization and prescription trends—and there are emerging methodologies for doing this—will be able to quantify the revenue at stake in the AI information environment.
Pharmacovigilance obligations. The cost of a significant pharmacovigilance failure—a safety signal that was visible in the AI information environment but not detected and reported—can be existential. The FDA can pull marketing authorization. Plaintiffs’ counsel will use evidence of awareness gaps to support punitive damages claims.
The investment required to build a systematic AI monitoring program—using tools like DrugChatter or building internal capability—is modest relative to any of these four risk categories. The companies that have built these programs report that the cross-functional value they produce—across regulatory, medical affairs, commercial, and legal—makes the ROI case internally more straightforward than most new regulatory compliance investments.
Where This Goes Next
The FDA’s Office of Prescription Drug Promotion is currently working on finalized guidance for AI-generated promotional content. Multiple people who participated in the agency’s 2024 public meetings on this topic describe an agency that is more attentive to this issue than its slow public output suggests. Finalized guidance is expected by 2026, and the expectation among regulatory attorneys advising major pharma companies is that it will include explicit monitoring requirements.
The AI platforms themselves are under pressure from multiple directions to improve the accuracy of their health-related outputs. Google DeepMind, OpenAI, and Anthropic have all announced health-related accuracy initiatives. Some of those initiatives include engagement with pharmaceutical companies about ensuring that their products’ prescribing information is accurately represented in AI outputs.
That engagement process is new and informal. There’s no established protocol for how a pharmaceutical company escalates an AI accuracy concern to a major AI provider, gets it reviewed, and verifies that it’s been corrected. The companies that have navigated this process—and a handful have—describe it as relationship-intensive, slow, and inconsistently effective. But it’s the current state of play, and companies that have built monitoring programs have at least identified the problem and begun the process.
The informational stakes will rise as AI tools become more capable of providing personalized health recommendations. The current generation of AI tools provides general information with disclaimers. The next generation—agentic AI tools that can access a patient’s health record, review their medication history, and provide personalized treatment recommendations—will function in a regulatory environment that is far more complex and consequential. Companies that build AI monitoring infrastructure now are building the organizational capability they’ll need when the stakes are higher and the regulatory environment is sharper.
Key Takeaways
- AI tools have become a primary health information layer for American patients, with roughly 40% consulting an AI tool around prescription decisions. That makes AI-generated content about drugs commercially and regulatorily consequential at a scale that most pharma monitoring programs weren’t built to address.
- The FDA’s OPDP has signaled—through warning letters, advisory communications, and public conference presentations—that AI monitoring is part of the same surveillance obligation that covers social media. Companies that treated social media monitoring as optional in 2014 learned an expensive lesson. The same trajectory is visible here.
- Three distinct risk categories make the investment case: regulatory enforcement exposure, litigation discovery exposure, and commercial market share impact from AI share of voice dynamics. All three are quantifiable and all three are growing.
- Effective AI monitoring requires systematic query frameworks built from patient language, coverage across all major AI platforms including AI-assisted search, regular sampling intervals rather than periodic audits, and documentation standards that meet regulatory defensibility requirements.
- The organizational model that works puts medical affairs in the lead—because accurate scientific representation is their core mission—with regulatory oversight and commercial teams as primary consumers of the intelligence output.
- Platforms like DrugChatter are filling a monitoring gap that neither traditional social listening tools nor internal pharmacovigilance systems were designed to address. The category is early enough that companies entering it now will set their own standards rather than having to catch up to competitors who moved first.
FAQ
Q: Does the FDA have the legal authority to require pharmaceutical companies to monitor AI outputs from third-party systems like ChatGPT or Gemini?
A: The FDA’s authority over pharmaceutical promotion derives from the FDCA and extends to any promotional activity where the manufacturer has a material connection—directly or indirectly. For purely third-party AI outputs with no manufacturer involvement, the agency’s direct enforcement authority is limited. But the constructive knowledge standard applied in adverse event reporting, the broader obligation to correct misbranding in the marketplace when a company has awareness of it, and the emerging pharmacovigilance expectations around digital channels collectively create a compliance environment where systematic monitoring is the only defensible position. The FDA rarely needs explicit statutory authority to make non-monitoring look like a bad idea in an enforcement action.
Q: How frequently should a pharmaceutical company’s AI monitoring program sample major AI platforms, and what triggers should prompt immediate review?
A: Monthly sampling is the minimum for a defensible program, with real-time alert capability for queries involving your brand name combined with specific safety terms, litigation-adjacent language, or off-label indication descriptions. Model updates from major AI providers—which can materially change output behavior—should trigger an immediate audit round. For drugs in active litigation, products with boxed warnings, or brands in competitive markets with high generic or biosimilar pressure, weekly sampling is more appropriate. The key is that the methodology is pre-defined and documented, not reactive.
Q: What recourse does a pharmaceutical company have when a major AI platform is consistently mischaracterizing a drug’s safety profile?
A: There is currently no formal regulatory mechanism for pharmaceutical companies to compel AI providers to correct drug information, unlike the established process for correcting misinformation in peer-reviewed literature. The practical pathway is relationship-based: most major AI providers have health policy or trust-and-safety teams that will engage with formally documented accuracy concerns from pharmaceutical companies, particularly when those concerns are substantiated with specific output captures, label references, and peer-reviewed evidence. Some companies have submitted formal accuracy requests and received corrections within weeks; others have submitted the same requests and seen no change six months later. Documentation of the attempt is valuable regardless of the outcome.
Q: How does AI monitoring data integrate with a pharmaceutical company’s existing pharmacovigilance infrastructure?
A: Most existing pharmacovigilance systems were designed to capture individual case safety reports from identifiable sources—physicians, patients, published case reports. AI monitoring data is aggregate and non-individual, which means it doesn’t map directly onto existing ICSR workflows. The most practical current integration is treating AI monitoring output as a signal-detection input: if AI monitoring identifies that a specific adverse event term is consistently associated with a drug in AI outputs, the PV team evaluates whether that association reflects signal-worthy information in the AI’s training corpus that warrants investigation through traditional PV channels. Some companies have built this as a formal process step; most have it as an informal escalation protocol. The FDA’s PV guidance doesn’t yet address this specifically, which gives companies latitude—and responsibility—to design their own approach.
Q: What is the competitive intelligence value of AI monitoring for drugs that are not yet commercially launched but are in late-stage clinical development?
A: Pre-launch AI monitoring is underused and genuinely valuable. For drugs in Phase 3 or NDA review, AI tools are already generating information based on published clinical data, conference presentations, and analyst coverage. Monitoring what AI says about a drug’s clinical profile before launch gives the brand team intelligence on how the information environment is characterizing the drug’s differentiation—which informs launch messaging, HCP education priorities, and competitive positioning before the company has any promotional presence. It also gives the regulatory team early visibility into mischaracterizations that need to be corrected before they become entrenched. For category-creating drugs entering a new indication, pre-launch AI monitoring can identify the patient query language that the brand team needs to address in its patient education strategy.