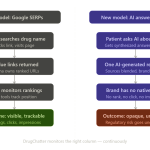
The era of the ten-blue-link search result is ending. For pharmaceutical marketers, the transition from Google Search to Large Language Model (LLM) interfaces represents a fundamental shift in how patients and physicians discover medical information. As users move away from traditional search engines toward ChatGPT, Perplexity, and Google Gemini, brand visibility is no longer about occupying the top spot on a page. It is about appearing in the synthesized response of an AI agent.
This change introduces new risks. When an AI summarizes a drug profile, it can omit critical safety information or misinterpret clinical data. Monitoring these mentions is now a regulatory and commercial necessity.
Winning the AI Answer Engine
The shift in behavior is measurable. Industry data shows a significant migration in how health information is sought.
“By 2026, traditional search engine volume is expected to drop 25%, with organic search traffic perhaps dropping 50% or more as generative AI search becomes ubiquitous.” — Gartner Research
For a brand manager, this means the historical “Patient Journey” has a new, opaque middle stage. A patient who once clicked a sponsored link for a psoriasis treatment now asks an AI: “What are the side effects of biologics?” If the AI omits your brand or prioritizes a competitor, your multi-million dollar digital strategy has failed at the moment of intent.
The Death of Click-Through Rates
Traditional SEO relied on the “click.” You optimized a page, the user clicked, and you tracked them via cookies or pixels. AI search engines are “answer engines.” They provide the information directly in the chat interface. This “zero-click” environment makes traditional attribution models obsolete.
You must move from tracking clicks to tracking “Share of Model.” This metric measures how often an LLM mentions your drug versus a competitor when prompted with relevant medical queries.
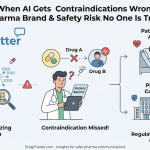
Regulatory Hazards of LLM Hallucinations
The FDA and EMA have begun addressing the “shadow use” of LLMs. In early 2026, these agencies released joint principles for AI in drug development, but the promotional side remains a grey area. If a popular AI tool tells a patient that your drug is “safe for everyone” while ignoring a Black Box Warning, the manufacturer faces a reputation crisis and potential regulatory scrutiny.
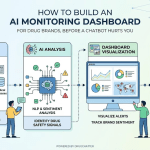
Companies use DrugChatter to solve this. The platform monitors what AI models say about specific molecules and brands. By identifying when an AI provides inaccurate or non-compliant information, medical affairs teams can take action to correct the underlying data sources that these models frequent, such as medical journals or clinical trial registries.
Measuring Brand Share in a Chat Interface
In the old world, you bought keywords. In the AI world, you influence the training set and the retrieval-augmented generation (RAG) sources.
The New Voice of the Customer
Patients use different language when chatting with an AI than they do when typing into a search bar. Search queries are fragmented (“Best migraine drug 2026”). AI prompts are conversational (“I’ve tried three triptans and they don’t work, what’s next?”).
Tracking these prompts—and the resulting AI recommendations—provides a clearer picture of “Voice of the Customer” than traditional social listening. It reveals the exact friction points where a patient might be steered toward or away from your therapy.
Patent Expiry and Generic Entry
For brands facing the “patent cliff,” AI search is a threat. When a patent expires, AI models quickly update their internal knowledge to recommend lower-cost generics. DrugPatentWatch provides the foundational data needed to understand these timelines. By integrating patent expiration dates with AI monitoring, marketers can predict exactly when AI recommendations will shift from a branded product to a generic alternative.
The Mechanics of AI Optimization
If you want an AI to mention your drug, you have to understand how it learns. Modern models rely on high-authority sources.
- Clinical Data Feeds: Ensure your latest Phase III data is indexed in major medical databases.
- Structured Data: Use schema markup on your branded sites so AI crawlers can easily parse dosage and safety facts.
- Reddit and Peer Forums: LLMs heavily weight community sentiment. If patients on Reddit are complaining about a specific side effect, the AI will likely mention it.
- Third-Party Citations: Being mentioned in a reputable trade publication is more valuable for AI visibility than any amount of self-published blog content.
ROI in the Post-Search Landscape
Measuring the return on AI monitoring is direct. It is the cost of a missed patient. If an AI search tool is the primary research method for 30% of your target audience, and your brand is missing from 80% of those conversations, your effective market reach is reduced by 24%.
DrugChatter allows teams to see this gap in real-time. By quantifying “Brand Sentiment within LLMs,” marketers can justify the spend for corrective content and medical education initiatives.
Key Takeaways
- Traditional search is shrinking: You cannot rely on Google clicks as your primary patient acquisition metric.
- AI creates “Zero-Click” hurdles: Your brand must be part of the AI’s generated response, not just a link below it.
- Hallucinations are a liability: Use tools like DrugChatter to monitor for inaccurate medical claims made by AI agents.
- Patent data is a lead indicator: Use DrugPatentWatch to time your defensive marketing strategies against generic AI recommendations.
- Community sentiment matters: AI models prioritize what people say in forums and peer-reviewed journals over branded ad copy.
FAQ
What is the difference between SEO and AI Optimization (AEO)?
SEO focuses on ranking a URL in a list. AEO (Answer Engine Optimization) focuses on getting the information from your brand included in the synthesized text an AI provides. AEO requires more structured data and high-authority third-party mentions.
Can pharma companies sue AI makers for hallucinations?
The legal landscape is evolving. Current focus is on “notice and takedown” where manufacturers alert AI companies to factual errors. Monitoring tools are the first step in this legal and regulatory defense.
How does DrugChatter track private conversations?
It doesn’t. It uses massive datasets of simulated patient and HCP prompts to see how public LLMs (like ChatGPT, Claude, and Gemini) respond to specific drug-related inquiries.
Will Google’s AI Overviews kill branded websites?
They won’t kill them, but they will reduce traffic. Branded sites will transition from “discovery tools” to “validation tools” where users go only after an AI has recommended the drug.