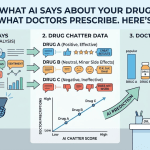
Scroll through enough patient forums and a pattern emerges: people aren’t just asking about side effects—they’re experimenting in real time. Mixing therapies, timing vaccines, layering supplements, or pushing dosing boundaries.
On DrugChatter, these questions often surface before clinicians ever hear about them. Some are harmless. Others sit squarely in high-risk territory—where off-label decisions, drug interactions, or misunderstood mechanisms can have real consequences.
Here are ten of the most dangerous off-label questions circulating right now—along with what makes them risky and why they matter.
1. Biologics + Live Vaccines: A Timing Gamble
Question: How long should I wait after cosentyx for live flu vaccine?
The combination of Cosentyx and live vaccines raises a fundamental immunology problem: suppression versus stimulation. Patients are effectively trying to time immune suppression against a vaccine that requires replication.
Risk signal: Potential for reduced vaccine efficacy—or worse, uncontrolled infection from the vaccine strain.
2. Dual NSAIDs: Doubling Up on Risk
Question: Can aspirin be taken with aleve?
Combining aspirin and Aleve looks harmless to patients seeking “stronger pain relief.”
Risk signal: This is a classic setup for gastrointestinal bleeding and renal stress—without proportional benefit.
3. Statins + Warfarin: A Narrow Therapeutic Window
Question: Lipitor and warfarin dosage concerns?
When Lipitor meets warfarin, small pharmacokinetic shifts can have outsized clinical effects.
Risk signal: Elevated bleeding risk due to altered anticoagulation levels—especially without monitoring.
4. Immunosuppressant Stacking
Question: Can you take cosentyx and methotrexate together?
Patients often assume “more treatment = better control.” Combining methotrexate with biologics like Cosentyx is sometimes clinically justified—but not casually.
Risk signal: Compounded immunosuppression increases infection risk and complicates monitoring.
5. Alcohol + Neuropathic Agents
Question: Can lyrica’s side effects be worsened by alcohol consumption?
Lyrica is already sedating. Adding alcohol introduces additive CNS depression.
Risk signal: Impaired coordination, respiratory depression, and increased accident risk—especially underreported in real-world use.
6. Grapefruit and Statins: The Enzyme Trap
Question: What interactions occur with lipitor grapefruit oil?
Grapefruit isn’t just food—it’s a CYP3A4 inhibitor. With Lipitor, that matters.
Risk signal: Elevated statin levels → increased risk of muscle toxicity, including rare but serious rhabdomyolysis.
7. Blood Pressure Collapse Concerns
Question: Can entresto cause low blood pressure?
Entresto is designed to lower cardiovascular strain—but patients often underestimate how far that effect can go.
Risk signal: Symptomatic hypotension leading to dizziness, falls, or fainting—especially in polypharmacy settings.
8. Vaccine Interactions with Biologics
Question: Does the covid vaccine interact with cosentyx?
Unlike live vaccines, mRNA vaccines (e.g., COVID-19 vaccines) don’t replicate—but immune modulation still matters.
Risk signal: Reduced immune response, leaving patients less protected than expected.
9. NSAIDs + Alcohol: A Quiet Hazard
Question: Are there any risks with mixing alcohol and advil?
Advil is widely perceived as “safe.” Alcohol changes that equation.
Risk signal: Increased gastrointestinal bleeding and liver stress—particularly with repeated use.
10. Herbal Supplements + Biologics
Question: Can I take herbal supplements while on cosentyx?
“Natural” doesn’t mean neutral. Herbal products can modulate immune pathways or drug metabolism.
Risk signal: Unpredictable immune effects or altered drug exposure—largely unstudied in combination with biologics.
What These Questions Reveal
Individually, these questions look like edge cases. Collectively, they point to something bigger:
- Patients are self-managing complex regimens without full visibility into interaction risk
- Off-label thinking is becoming normalized, especially in chronic disease communities
- Food, supplements, and lifestyle factors are increasingly treated as interchangeable with drugs
In short: the boundary between prescribed therapy and experimental behavior is blurring.
Why This Matters for Industry
For pharmaceutical companies, regulators, and data providers, this is not just anecdotal noise. It’s early warning signal data:
- Emerging off-label combinations before they hit claims databases
- Real-world adherence distortions (timing, stacking, substitution)
- Safety signals forming outside controlled environments
Platforms like DrugChatter are effectively front-line pharmacovigilance feeds—if you know how to read them.
Bottom Line
The most dangerous off-label use isn’t happening in clinical trials—it’s happening in everyday decisions:
- Taking two drugs instead of one
- Adding a supplement without disclosure
- Timing a vaccine based on guesswork
Each question above represents a patient trying to solve a problem. But without guidance, some of those solutions create new ones.
And increasingly, those risks show up first in places like DrugChatter—not the clinic.